Alright, buckle up, buttercups, because we're about to dive into a world that's more magical than a unicorn doing the cha-cha! We're talking about sulfur, that bright yellow stuff that looks like it was sprinkled by a mischievous fairy. Now, you might think sulfur is just… well, sulfur. Solid, grumpy, and hanging out in its solid state like it’s perpetually at a stuffy tea party. But oh, my friends, sulfur has a secret life, a secret transformation that’s way cooler than you can imagine. It's called sublimation, and it’s basically the ultimate magic trick in the science world!
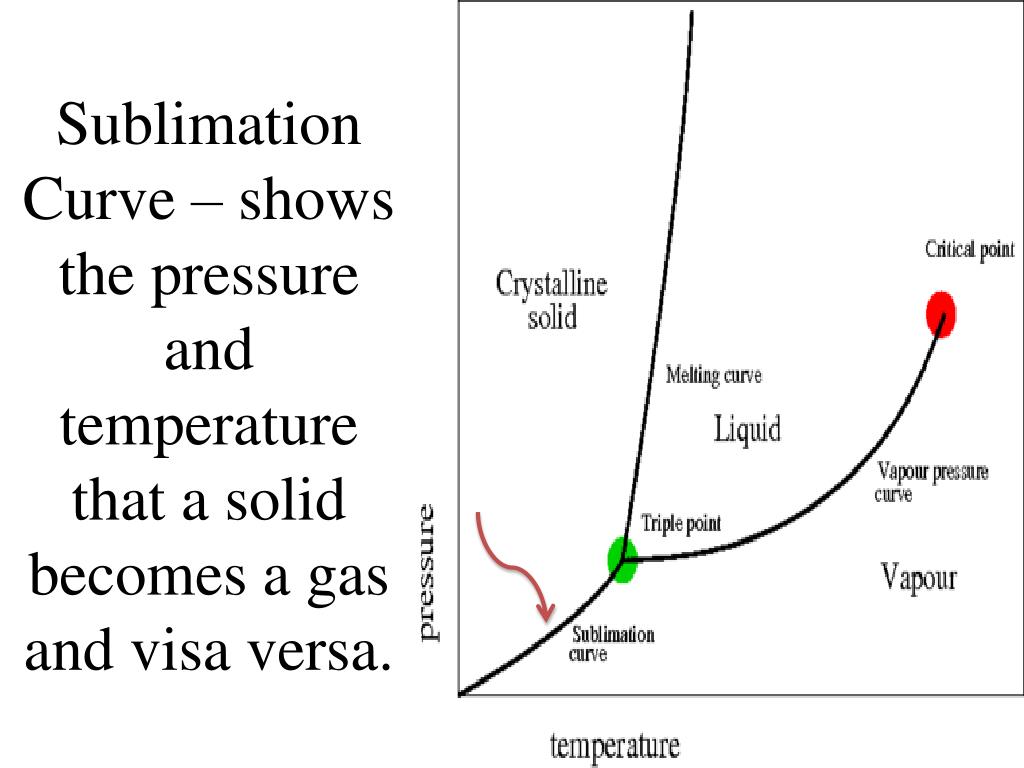
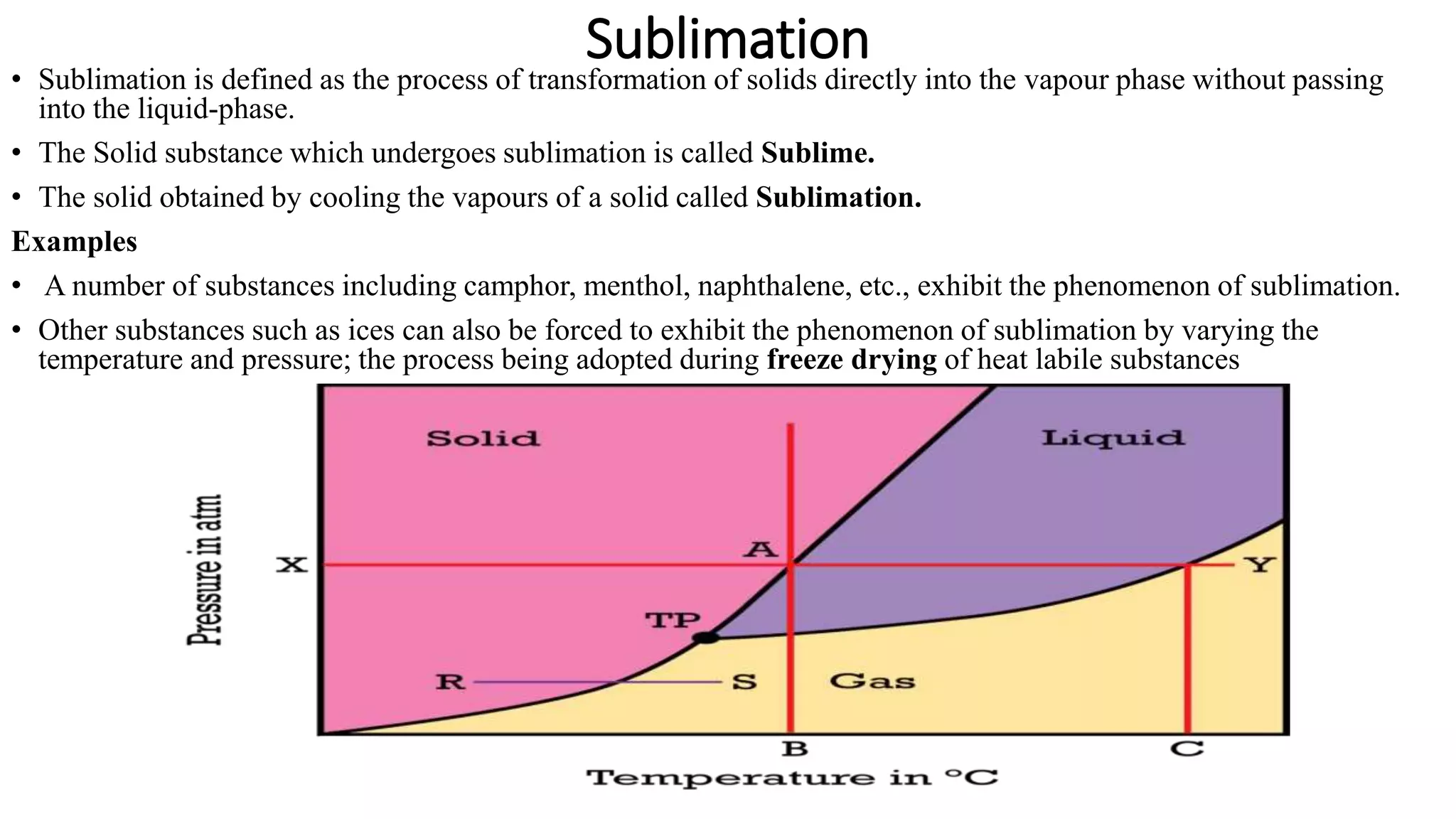
Imagine this: you’ve got a block of ice, right? Everyone knows ice melts into water. Pretty standard stuff. But imagine if, instead of melting, that ice just… poof! Vanished into thin air, becoming invisible water vapor right before your eyes! That’s essentially what sublimation is. It’s skipping the whole liquid phase and going straight from solid to gas. And our star player, sulfur, is a total pro at this particular disappearing act.
Now, you might be thinking, "Okay, so sulfur can do a magic trick. Big deal. My toaster does a magic trick every morning by turning bread into toast." And you'd be right! But here's where it gets really exciting. This sublimation party doesn't happen just anywhere. It has a VIP guest list, and the most important name on that list is pressure. Think of pressure like the bouncer at the coolest club in town. If you don't meet the bouncer's requirements, you're not getting in.
For solid sulfur, this "pressure bouncer" is incredibly chill. Like, ridiculously chill. It's way more laid-back than that grumpy bouncer at your local dive bar.
We’re talking about a pressure so low, it’s practically non-existent. Imagine the quietest whisper you’ve ever heard. Now imagine something even quieter. That’s the kind of pressure we’re talking about. It’s the kind of pressure you might find way, way up in the clouds, like where the fluffy unicorns nap, or even higher, in the vast, silent expanse of outer space. Seriously, it’s that low!
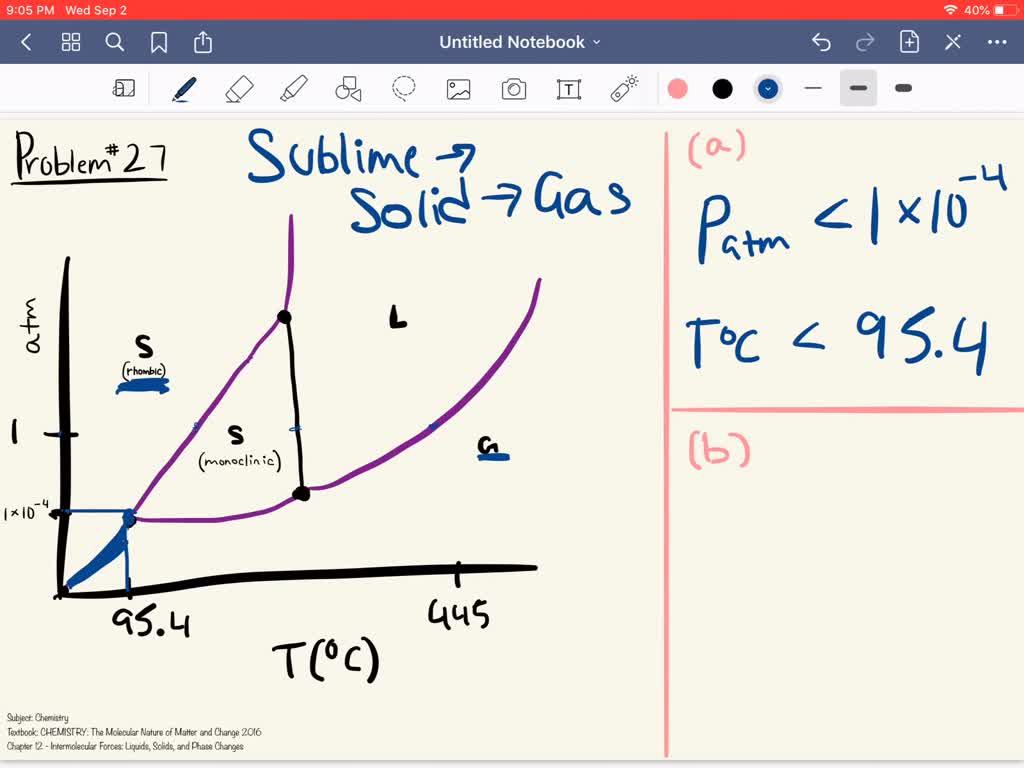
So, what's the magic number? Below what pressure will solid sulfur decide to ditch its solid suit and embrace the gaseous life? Drumroll, please! It’s somewhere around 0.00001 millimeters of mercury. Yes, you read that right. That’s a decimal point followed by four zeros and then a one! If you were trying to measure that with a regular ruler, you’d be looking for a speck of dust that’s even smaller than a gnat’s sneeze.
Let’s put that into perspective. The pressure at the top of Mount Everest, which already feels pretty thin to us Earthlings, is about 250 millimeters of mercury. That’s like comparing a full-on thunderstorm to a single, shy little raindrop. Our sulfur sublimation pressure is so minuscule, it’s like asking if you can hear a butterfly clearing its throat in a hurricane. It’s that drastic a difference!
So, what does this mean for our dazzling sulfur? It means that in environments where the pressure is super, super low, our yellow friend doesn't need any extra heat to start its transformation. It's so eager to become a gas, it’ll do it just because the pressure is low enough. It’s like it’s been waiting its whole life for this moment, a chance to break free from its solid shackles and float around in the atmosphere like a free spirit.
Think about it: if you had a tiny bit of sulfur in a jar and you pumped all the air out until it was practically a vacuum, that sulfur would start to sublime all on its own. It would slowly but surely turn into a gas, leaving behind… well, maybe nothing! It would just disappear into the invisible, becoming part of the ethereal dance of gases. It's a silent, beautiful transformation that happens when the universe gives it the perfect conditions.
This isn't just some quirky science fact to impress your friends at parties (though it’s definitely good for that!). This ability of sulfur to sublime at such low pressures has some really cool implications. It means that in certain geological settings, or even in the atmosphere of other planets, sulfur could be doing this disappearing act all the time. It's like a hidden chemical ballet happening all around us, or even far, far away from us.
So, the next time you see or hear about sulfur, remember its secret superpower. Remember that beneath all that solid yellow goodness lies a gas waiting to happen, a molecule eager to break free. And it doesn’t need much encouragement, just a tiny, almost imperceptible drop in pressure. It’s a reminder that even the most seemingly ordinary things can have extraordinary abilities, and that the universe is full of quiet magic, just waiting for us to notice.
It’s a wonderfully humbling thought, isn’t it? That something as seemingly simple as a change in pressure can unlock such a dramatic transformation. It makes you wonder what other secrets the world is holding, what other magical transformations are just waiting for the right conditions to reveal themselves. And for that, my friends, we can all feel a little bit happier and a lot more entertained by the wonders of science!