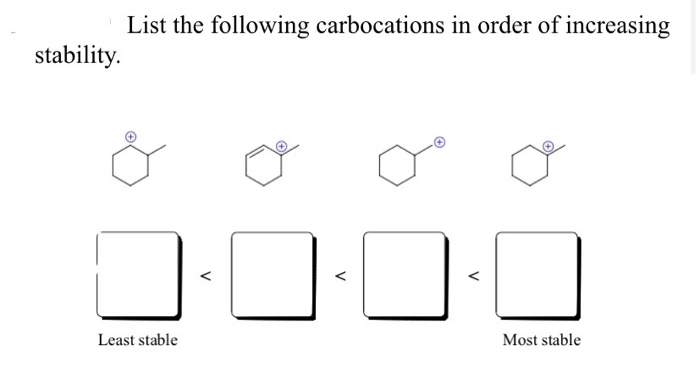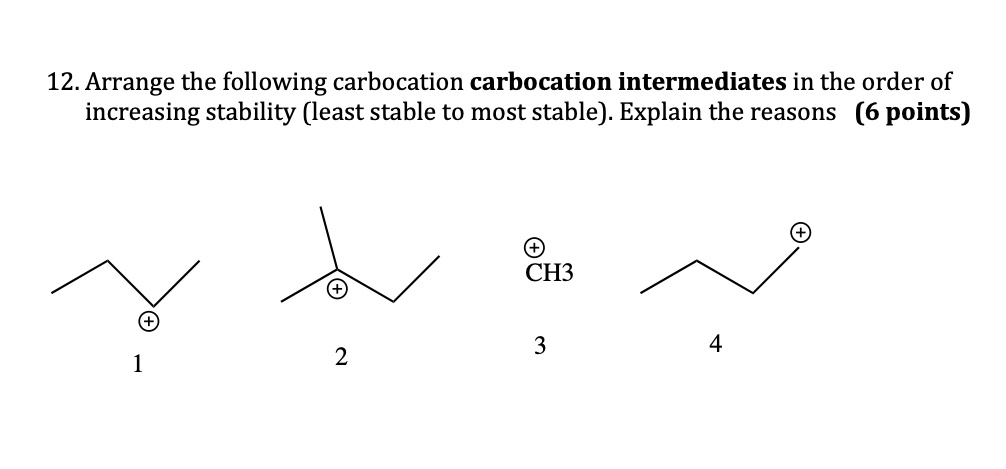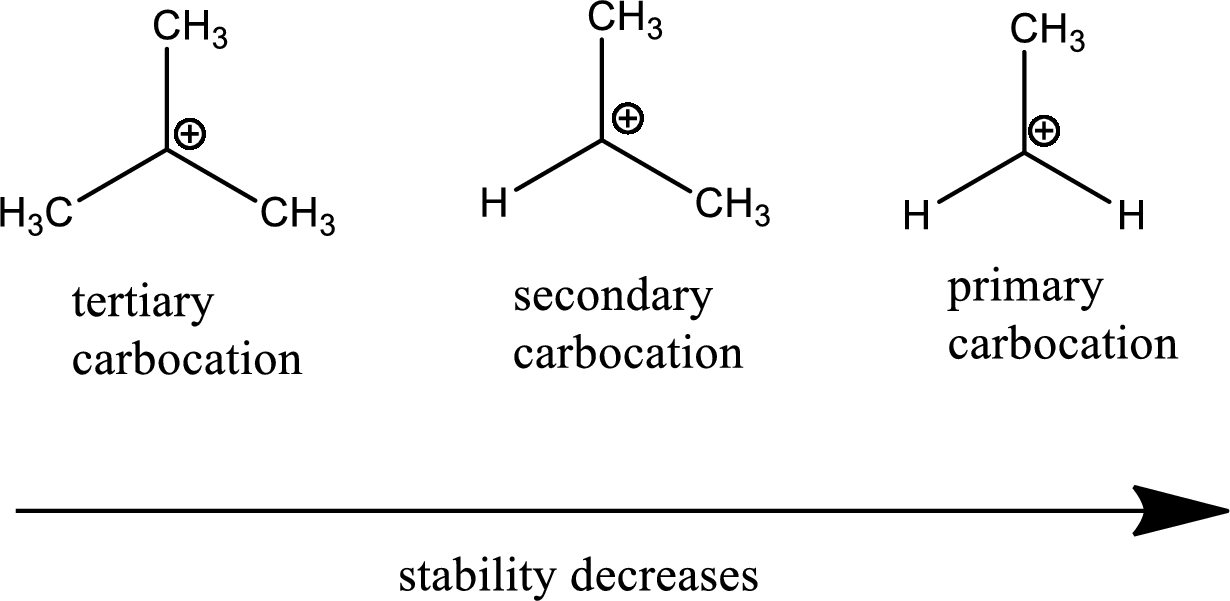
Hey there, fellow science nerds and curious cats! Ever wondered about the tiny, zippy things that make chemical reactions happen? Well, today we're diving into the wacky world of carbocations. Don't let the fancy name scare you. Think of them as really, really hungry little guys. They're basically carbon atoms that have lost an electron and are feeling a bit… positively charged and desperate for some company. Like that one friend at a party who’s always looking for a snack. You know the type!
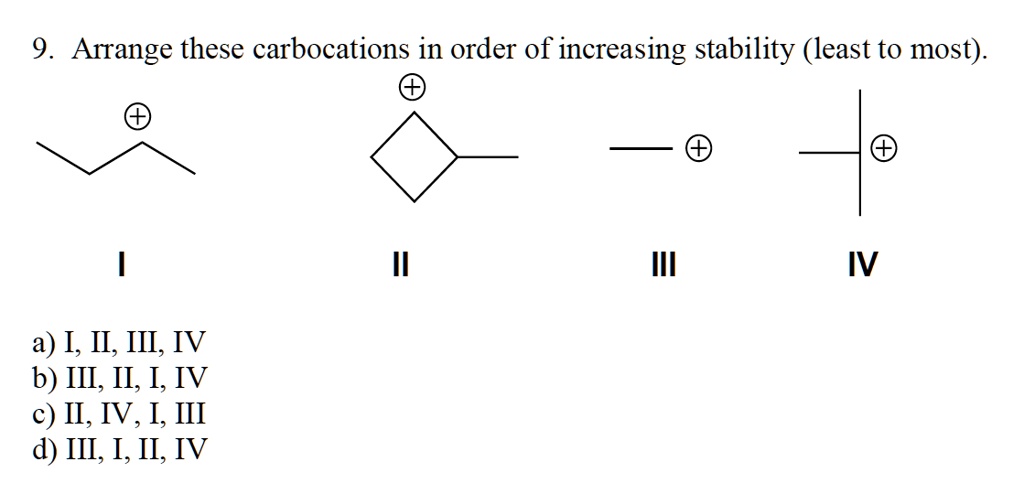
And guess what? Just like us humans, these carbocations have their own little social hierarchy. Some are super chill, while others are practically vibrating with need. We're talking about their stability. Which ones are the most popular? The ones that can handle the pressure, you know? We're going to explore this order, from the least stable to the most, and it’s surprisingly more fun than it sounds. Get ready for some chemistry giggles!
The Unstable Wannabes: Methyl and Primary Carbocations
Let's kick things off with the least stable of the bunch. Meet the methyl carbocation. It’s like the shy kid in the back of the class. A single carbon atom with a positive charge and just three hydrogens hanging around. It’s not exactly the life of the party. It’s so needy, it’ll react with pretty much anything just to get some electrons. Imagine trying to have a conversation with someone who interrupts you every two seconds because they’re desperate for attention. Yeah, that’s our methyl carbocation.
Then we have the primary carbocation. Think of it as the methyl carbocation with a tiny upgrade. It’s still a bit wobbly, though. We’re talking a carbon with a positive charge, attached to one other carbon group, and the rest are hydrogens. It’s like your friend who’s a little bit more confident than the shy one, but still needs a lot of reassurance. They’re still pretty eager to grab those electrons and settle down. They're like the person who almost gets the joke but needs a little nudge.
Why are they so unstable? Well, the positive charge on the carbon is a real bummer for it. It wants to be neutral, like everyone else. And these guys don't have much help from their neighbors. It’s like trying to balance a teeter-totter with only one person on the other side. Not very stable, right? They’re just waiting for something to come along and make them feel whole again. Poor things!
A Quirky Fact: The "Electron Deficiency" Tango
It's all about being electron deficient. Carbon loves to have eight electrons in its outer shell, like a cozy, complete family. But carbocations are missing one! They're like a puzzle with a missing piece. So, they're constantly on the lookout for more electrons to complete their happy little octet. It’s a never-ending quest for electron buddies!
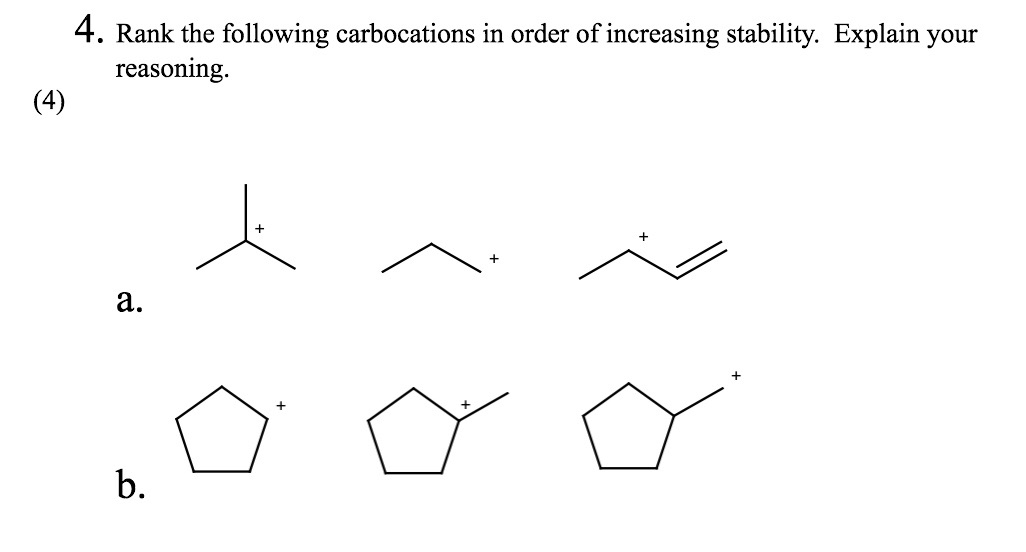
The Middle Ground: Secondary Carbocations
Alright, let’s move up the ladder of stability. Enter the secondary carbocation. Now we’re talking! This little guy has a positive charge on a carbon atom that’s attached to two other carbon groups. Think of it as the friend who’s got a couple of buddies backing them up. They’re not as desperate as the methyl or primary ones. They have a little more support.
Why are they more stable? Ah, this is where things get interesting. Those neighboring carbon groups are actually pretty helpful. They have these things called alkyl groups, which are like little electron-donating buddies. They can push a bit of their electron density towards the positively charged carbon. It’s like having a few friends in the room who can distract you from your problems. They’re not solving everything, but they’re definitely making things a bit more bearable.
This effect is called hyperconjugation. Sounds fancy, right? But it’s basically just the neighboring carbon atoms sharing their electron love. They’re not giving it all away, mind you, but they’re offering a little bit of comfort. It's like a gentle pat on the back. Enough to make you feel a bit better, but you still have that positive vibe going on.
So, compared to the primaries, secondary carbocations are a bit more chill. They’re still looking for a reaction, but they can wait a smidge longer. They've got a bit more swagger. They’re not sprinting to the nearest electron source; they might even stroll. A slight improvement, but we’re not at the top tier yet!
A Funny Thought: The "Alkyl Group Hug"
Imagine the neighboring alkyl groups giving the positively charged carbon a big, comforting “alkyl group hug.” They’re not squishing it, just providing a little bit of warmth and support. It’s the chemical equivalent of a reassuring squeeze. Adorable, isn't it?
The Rockstars of Stability: Tertiary Carbocations
And now, drumroll please… the undisputed champions of carbocation stability: the tertiary carbocations! These are the divas, the celebrities, the ones everyone wants to be around. A tertiary carbocation has a positively charged carbon atom that’s attached to three other carbon groups. Three! That’s a whole entourage!
Why are they so incredibly stable? It’s all about that electron donation power. Those three neighboring alkyl groups are really good at sharing their electron love. They’re practically showering the positively charged carbon with electron density. It's like having a whole cheering squad yelling encouragement. They’re taking all that positive energy and diffusing it, making the carbocation much more relaxed and less likely to freak out.
The hyperconjugation here is off the charts! With three alkyl groups, there are way more opportunities for those neighboring C-H bonds to share their electrons. This dramatically stabilizes the positive charge. It’s like having a security detail for your positive vibes. You’re just not as vulnerable.
Tertiary carbocations are the coolest cats in the reaction world. They can hang out for a bit longer without causing a fuss. They’re the ones that are more likely to be found in reactions that require a bit more time and patience. They’re the chill ones who can handle the heat. They’ve got it all figured out, at least as much as a carbocation can!
A Quirky Detail: The "Positive Charge Diffusion Dance"
Think of it like a really organized dance party. The three alkyl groups are leading the positively charged carbon in a smooth, coordinated dance that spreads out the positive charge, making it less intense and therefore more stable. It's a beautiful, electron-sharing ballet!

The Grand Finale: Why Does This Even Matter?
So, why all this fuss about who’s more stable? Well, understanding this order is super important in chemistry. It helps us predict how reactions will happen. The more stable a carbocation is, the more likely it is to form. It’s like knowing which party is going to be the most fun – you’re going to aim for that one, right?
This knowledge is key for predicting reaction products, understanding reaction mechanisms, and even designing new chemical processes. It’s the backbone of a lot of organic chemistry. Without knowing which carbocation is king, we’d be lost in a sea of unpredictable reactions. It’s like trying to navigate without a map!
It's a fundamental concept, but it has these quirky, almost personified aspects that make it fun to think about. These little charged carbon fragments have their own dramas, their own hierarchies. It’s a microscopic soap opera playing out in every chemical flask. And honestly, who doesn't love a good drama?
So next time you hear about carbocations, remember their little stability dance. From the needy methyl to the cool-as-a-cucumber tertiary, they’ve got a whole spectrum of personalities. It's a reminder that even in the world of atoms and molecules, there’s a fascinating order and a whole lot of fun to be had if you just look close enough!