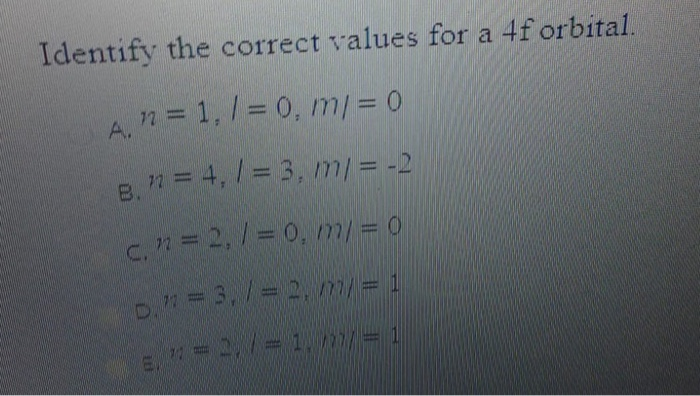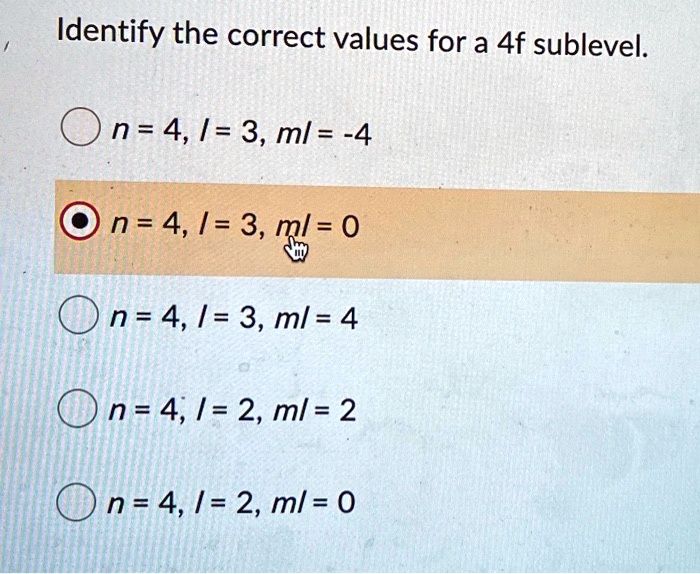
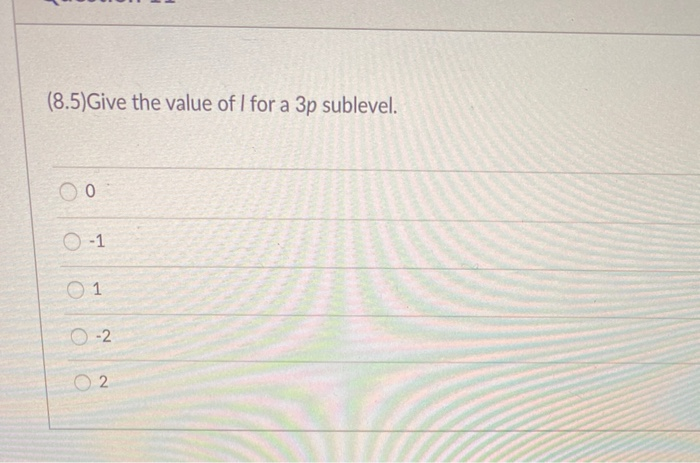
Ever wondered about the tiny, intricate world of atoms? It's a place brimming with fascinating structures, and today we're going to peek into one of its most intriguing corners: the 4f sublevel. Now, that might sound a bit technical, but stick with me, because understanding it can be surprisingly fun and surprisingly relevant to things you might encounter every day, even if you don't realize it!
Think of an atom like a miniature solar system, with electrons orbiting the nucleus. These orbits aren't just random; they're organized into different "energy levels" and, within those, even more specific "sublevels." The 4f sublevel is a particular type of electron housing, and understanding its correct values helps us predict and explain a whole lot of chemical behavior. Why is this important? Well, it's like knowing the rules of a game. Once you know the rules (the correct values), you can understand why certain players (elements) behave the way they do.
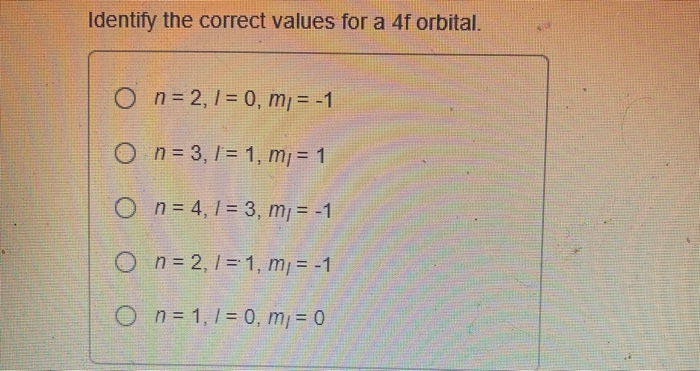
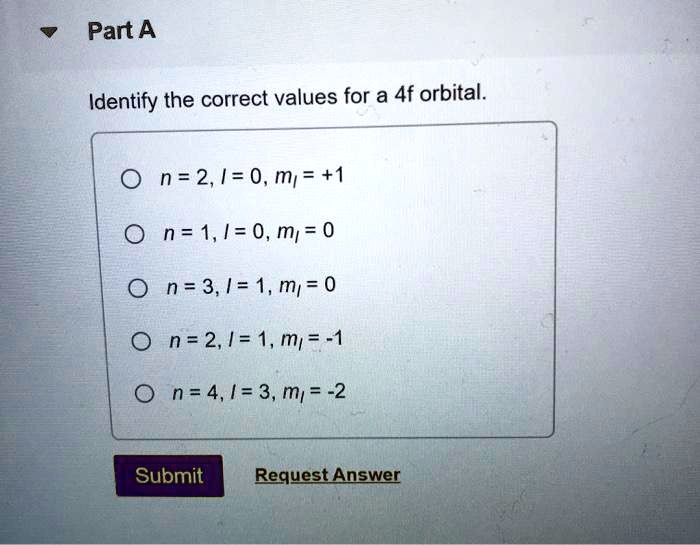
The main purpose of understanding the 4f sublevel is to accurately describe the electron configurations of atoms, especially those in the lanthanide series. These elements, often found in magnets, lasers, and even color TV screens, have their outermost electrons residing in or influencing the 4f sublevel. By knowing the correct values associated with this sublevel – things like its energy, shape, and the number of electrons it can hold – scientists and students can predict how these elements will react, form bonds, and exhibit their unique properties.
So, where do you see this playing out? In education, it's a cornerstone of understanding chemistry and physics. Textbooks delve into quantum numbers and orbital shapes precisely because they govern how atoms interact. In daily life, the properties of the 4f elements, like their ability to absorb and emit light, are fundamental to technologies like LEDs and the phosphors that make your computer screen glow. Even the strength of the magnets in your refrigerator or your headphones often relies on the unique magnetic properties derived from elements whose electrons are shaped by the 4f sublevel.
Now, you might be thinking, "This sounds complicated!" But exploring it doesn't have to be a chore. A simple way to start is by looking at a periodic table. Notice the block of elements at the bottom, usually labeled as lanthanides and actinides? Those are the elements where the 4f sublevel plays a starring role. You can also search online for "electron configuration" or "atomic orbitals" to see visual representations of what these sublevels look like – they're quite intricate and beautiful in their own way. Don't be afraid to use visual aids! Seeing diagrams of the f-orbitals can make abstract concepts much more concrete. Think of it as a little mental puzzle, and the correct values are the pieces that fit together to reveal the atom's true character.
![[SOLVED] 10) (3 pts.) Identify the correct values for a 4f sublevel](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/12/6398f1a03e435_7126398f1a0157df.jpg)