Hey there, science adventurer! Ever looked at something super tiny, like a building block of everything, and wondered, "How on earth does that thing even hold together?" Well, today, we're diving into a concept that makes chemistry way more fascinating and, dare I say, even a little bit magical: hybridization in small molecules!
Now, don't let the fancy word scare you. Hybridization is just a super cool way chemists explain how atoms in a molecule decide to get cozy and form bonds. Think of it like atoms having a little chat and saying, "Okay, this original orbital shape isn't quite working for us to connect with our friends. Let's mix and match a bit to create something better!"
The Secret Life of Atomic Orbitals
So, what are these "orbitals" we're talking about? Imagine electrons aren't just little dots zipping around. They actually hang out in these fuzzy, three-dimensional regions around the atom's nucleus. These regions are called orbitals, and they have different shapes – like spheres (s orbitals) and dumbbells (p orbitals). Think of them as electron "neighborhoods."
Now, for a long time, scientists looked at simple molecules like methane (you know, the stuff that makes up natural gas) and thought, "Hmm, the way carbon is bonded to those four hydrogen atoms doesn't quite fit the simple s and p orbital shapes." It was like trying to fit square pegs into round holes, but for atoms!
This is where the brilliant idea of hybridization swoops in to save the day. It's like the atom's way of saying, "Hold on a minute, let's rearrange our electron neighborhoods to make a perfect fit!"
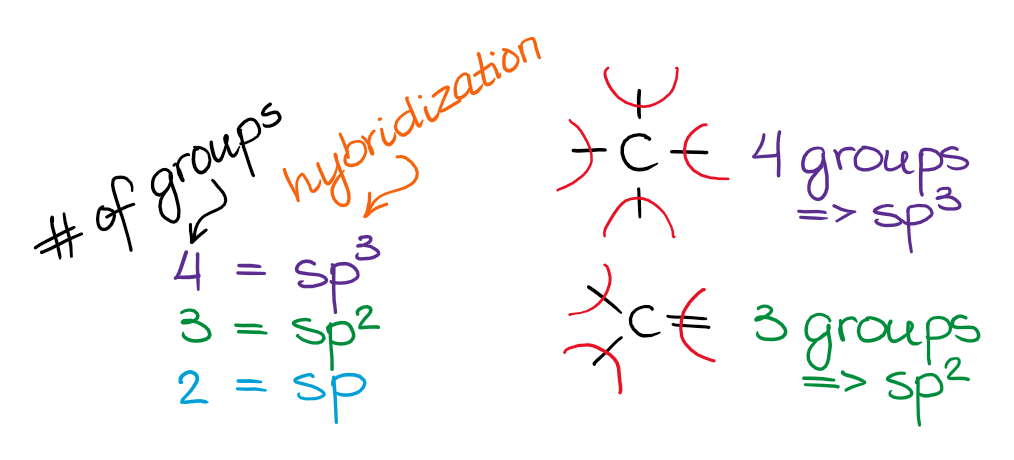
Mixing It Up: The Art of Sp3
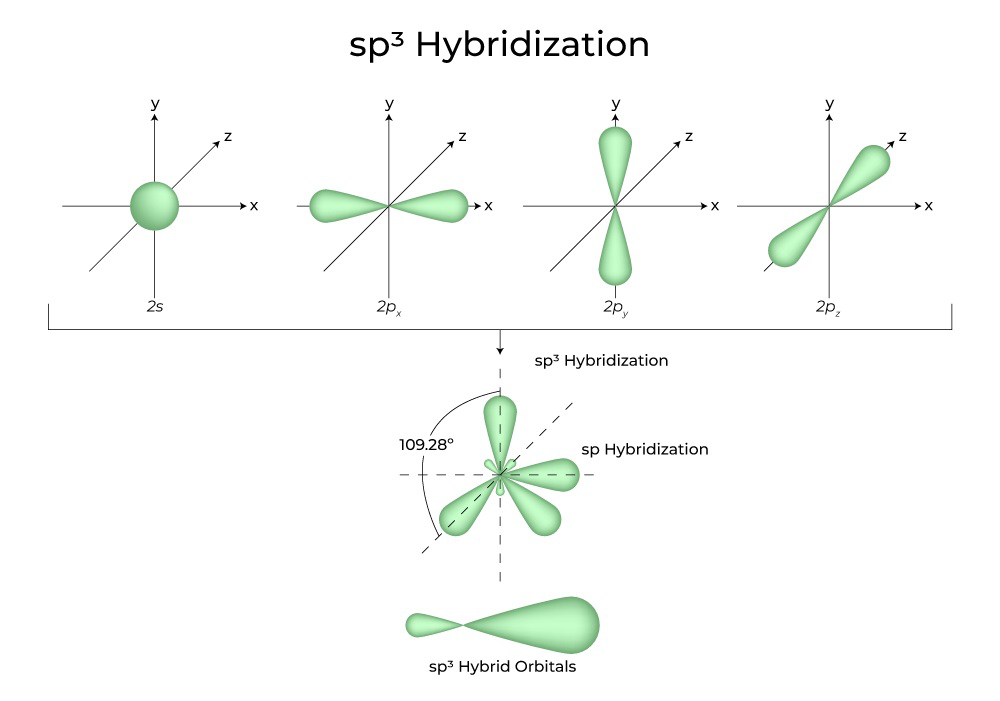
Let's take our methane (CH4) example. Carbon, bless its heart, usually has its electrons in one s orbital and three p orbitals. If it tried to bond with four hydrogens using just those original shapes, the bonds would all be different and at weird angles. But we know that in methane, all four C-H bonds are exactly the same length and strength, and they form a beautiful, symmetrical shape called a tetrahedron. How do we get there?
Enter sp3 hybridization! Imagine the carbon atom taking its one s orbital and its three p orbitals and shuffling them all together. It's like a blender for electron clouds! When it's done, it creates four brand new, identical orbitals. These new orbitals are called sp3 hybrid orbitals. They're a little bit s-like and a little bit p-like, and they're perfectly shaped and angled to point towards the corners of a tetrahedron.
This is why methane is so stable and why carbon can form so many amazing compounds. It's all thanks to this clever atomic reshuffling!
..jpg)
What About Double and Triple Bonds?
But wait, there's more! Hybridization isn't just for single bonds. It gets even more exciting when we look at molecules with double and triple bonds, like ethene (C2H4) which has a double bond between the two carbons, or ethyne (C2H2) with a triple bond.
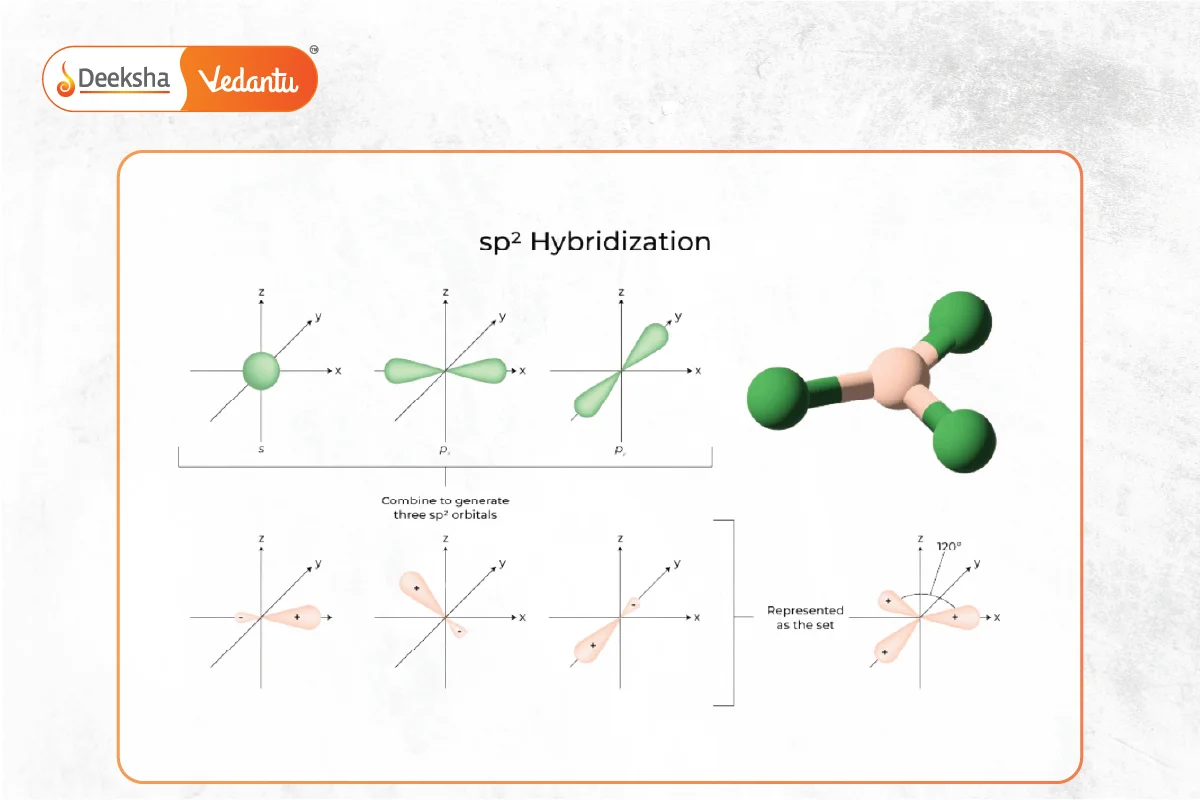
For ethene, where each carbon is bonded to two hydrogens and another carbon with a double bond, we see sp2 hybridization. Here, the carbon atom mixes one s orbital with two p orbitals. This creates three sp2 hybrid orbitals that lie in a flat plane, perfect for forming sigma bonds (the strong, direct bonds). But what about that double bond? The leftover p orbital on each carbon atom is crucial! It sticks out above and below the plane and overlaps side-by-side with the other carbon's leftover p orbital, forming a pi bond. This pi bond is like the "glue" that makes the double bond so special!
And for ethyne, with its triple bond? That's where sp hybridization comes in. The carbon atom mixes one s orbital with one p orbital, creating two sp hybrid orbitals that point in opposite directions. These form the sigma bond. The two remaining p orbitals on each carbon are then used to form two pi bonds. That's right, two!

Why Does This Make Life More Fun?
Okay, so it's a neat trick atoms play. But how does this make your life more fun? Well, understanding hybridization is like unlocking a secret code to how the world works! It's the foundation for understanding why water molecules behave the way they do, why your food tastes like it does (thanks to complex organic molecules!), and even how your body builds and repairs itself.
Think about it: the shapes of molecules, dictated by their hybridization, determine everything from whether a drug can fit into a receptor in your body to why a particular plastic is flexible or rigid. It's literally the shape of things that makes them do what they do!
When you start to grasp hybridization, you see the elegance and logic behind chemistry. It's not just random reactions; it's atoms cleverly arranging themselves to create the incredible diversity of matter around us. It turns a seemingly simple molecule into a fascinating puzzle of bonding and geometry.

A Little Spark of Inspiration
So, the next time you hear about a molecule, whether it's the caffeine that gives you that morning boost or the oxygen you breathe, remember the incredible atomic dance of hybridization happening within it. It's a testament to nature's ingenuity and the beautiful simplicity hidden within complexity.
You don't need to be a rocket scientist to appreciate this stuff! A little curiosity goes a long way. This is just a tiny peek into the amazing world of molecular structure. There's so much more to explore, so many more molecules waiting to reveal their secrets. Maybe this little chat has sparked a fire in you? Perhaps you'll find yourself wondering about the hybridization in the next molecule you encounter. And that, my friends, is a truly exciting journey to begin!
Keep asking questions, keep wondering, and keep discovering. The universe, down to its smallest building blocks, is an endlessly inspiring place!
.jpg)