Hey there, chemistry curious cats! Ever wondered what’s really going on inside those atoms you learned about in school? Like, what makes some of them all clingy and others a bit more aloof? Well, get ready, because we’re about to dive into the fascinating, and dare I say, electrifying world of effective nuclear charge. Don’t worry, it’s not as scary as it sounds! Think of it like a popularity contest for electrons, with the nucleus as the ultimate judge. And the prize? The electron’s affection (or, you know, just staying put in its orbit). So, grab a cuppa, settle in, and let’s unravel this mystery together!
So, what exactly is this “effective nuclear charge” thing? Imagine the nucleus of an atom is like a super-popular celebrity. It’s got a bunch of positive protons, which are like the paparazzi, all eager to get attention. But then you have the electrons – these are the adoring fans. Some fans are right up front, getting all the attention. Others are a bit further back, maybe behind a few other fans or even some security guards. The effective nuclear charge is basically how much of that celebrity’s (nucleus’s) charm and attention the outermost fans (electrons) actually feel. It’s the real pull they experience, not just the total power of the celebrity.
Why should we care about this? Ah, my friend, this is where the magic happens! Effective nuclear charge is the secret sauce that explains a ton of chemical behavior. It dictates how easily an atom will grab another electron, how much energy it takes to snatch one away, and even how big the atom will be. It’s like the underlying personality of the atom, shaping all its interactions.
Let’s break down the players. We’ve got the nucleus, with its positive protons. Then we have the electrons. Now, here’s the twist: electrons aren’t just chilling in their own little orbits, oblivious to each other. Oh no! They’re a bit of a social bunch. The inner electrons, the ones closer to the nucleus, are like the super-fans in the front row. They do a pretty good job of shielding, or blocking, the pull of the nucleus from the outer electrons. Think of it like trying to get a clear view of a celebrity from the back of a crowded stadium. Those people in front are definitely getting in the way!
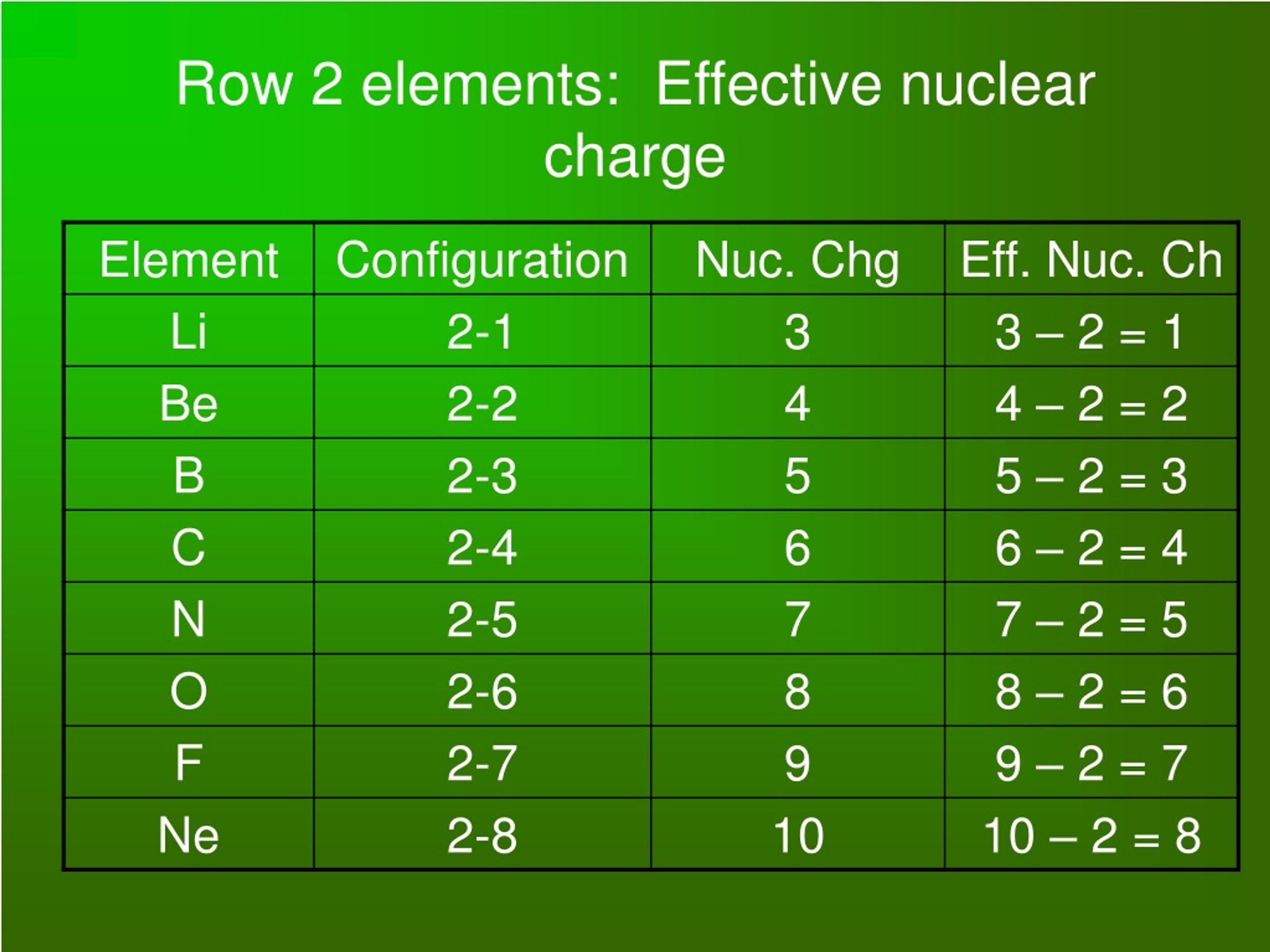
So, the effective nuclear charge (which we chemists often abbreviate as Zeff, fancy, right?) is the actual nuclear charge (the total number of protons, Z) minus the shielding effect of the inner electrons (S). So, Zeff = Z - S. It’s like the net amount of “oomph” the outer electrons feel. If S is big, Zeff is small. If S is small, Zeff is big. Simple as that! Well, almost simple. The electron shielding isn’t a perfect science, but this gives us a good grasp.
The Grand Ranking: Who's Got the Most "Zest"?
Alright, enough theory! Let’s get to the fun part: ranking these elements! When we talk about ranking by effective nuclear charge, we’re primarily looking at how this charge changes as we move across the periodic table. Think of it as a journey, a grand tour of atomic personalities.
Across a Period: The Popularity Contest Heats Up!
Let’s start with moving from left to right across a row, or a period, on the periodic table. This is where things get really interesting. As you move across a period, say from Lithium (Li) to Neon (Ne), the number of protons in the nucleus increases. So, Lithium has 3 protons, Beryllium has 4, Boron has 5, and so on, all the way to Neon with 10 protons. More protons means a stronger positive pull, right?

But wait, there’s a catch! While the number of protons is going up, the electrons are being added to the same outermost energy shell. This means the inner electron shells, the ones doing the shielding, aren’t really changing much. They’re doing their job, but they’re not getting any more of them to do a better job of shielding. It’s like the same security detail trying to manage an increasingly famous celebrity.
So, what happens? That increasing nuclear charge starts to win the battle against the relatively constant shielding. The outermost electrons feel a stronger and stronger pull from the nucleus. This means the effective nuclear charge increases as you move from left to right across a period. The celebrity is getting more and more popular, and the outer fans are feeling that intense adoration more directly!
Let’s take an example: Lithium (Li) is in Group 1, Period 2. It has 3 protons, and 2 inner electrons shielding the one outer electron. So, its Zeff is roughly 3 - 2 = 1. Pretty chill. Now look at Fluorine (F) in Group 17, Period 2. It has 9 protons, and 8 inner electrons shielding the 7 outer electrons. Now, those 8 inner electrons are doing a decent job of shielding, but not perfectly. So, the Zeff for Fluorine is much higher than for Lithium. It’s feeling a much stronger tug towards the nucleus. Fluorine is like the rockstar everyone’s dying to get a glimpse of!
So, the order across a period would look something like this (simplified, of course, but you get the gist): Group 1 < Group 2 < Group 13 < Group 14 < Group 15 < Group 16 < Group 17 < Group 18. The noble gases at the end are practically glued to their nuclei because of that high Zeff! They’re so stable, they often don’t bother interacting with anyone else. Very exclusive parties.
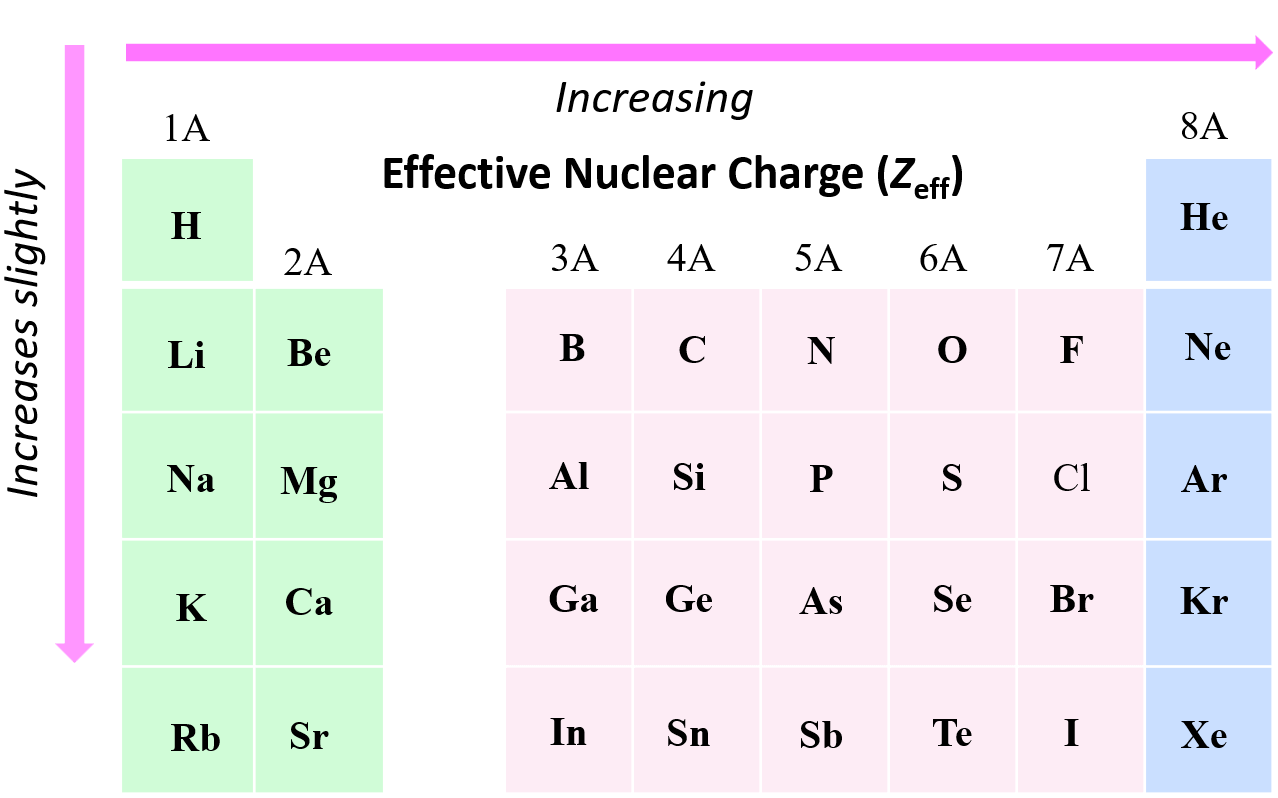
Down a Group: The "Fame" Distributes Differently
Now, let’s talk about moving from top to bottom down a column, or a group. This is where the story changes a bit. As you go down a group, say from Hydrogen (H) to Lithium (Li) to Sodium (Na), the number of protons in the nucleus increases. Hydrogen has 1 proton, Lithium has 3, Sodium has 11, and so on. That’s a bigger celebrity!
However, and this is the crucial part, you are also adding entirely new electron shells. Each new period you move down means a whole new layer of electrons is added, and these new shells are further away from the nucleus. Think of it as adding more and more layers of security and fans between the celebrity and the outermost fans. These new shells, and the extra inner electrons they contain, do a much better job of shielding the outermost electrons from the nucleus’s pull.
So, even though the number of protons is increasing significantly, the increase in shielding from the new electron shells largely cancels out that effect. The outermost electrons are still being pulled, but the pull is less concentrated. They’re feeling the celebrity’s fame, but it’s more diffused, like a distant roar rather than a personal shout-out.
Therefore, as you move down a group, the effective nuclear charge generally stays relatively similar, or increases only slightly. It’s not the dramatic increase we saw across a period. The number of protons goes up, but the shielding goes up even more, so the net pull on the outermost electrons doesn’t skyrocket. The celebrity is getting more famous, but there are also a lot more people between them and the people trying to get their attention.

So, within a group, the difference in Zeff isn’t as pronounced as it is across a period. Lithium (Period 2, Group 1) has an outer electron in the n=2 shell. Sodium (Period 3, Group 1) has its outer electron in the n=3 shell. That extra shell in Sodium provides significantly more shielding. While Sodium has more protons, those extra inner shells are doing a fantastic job of diluting the nucleus’s effect on that outermost electron.
Putting it All Together: The Big Picture
So, if we were to rank elements based on their effective nuclear charge, here’s the general trend:
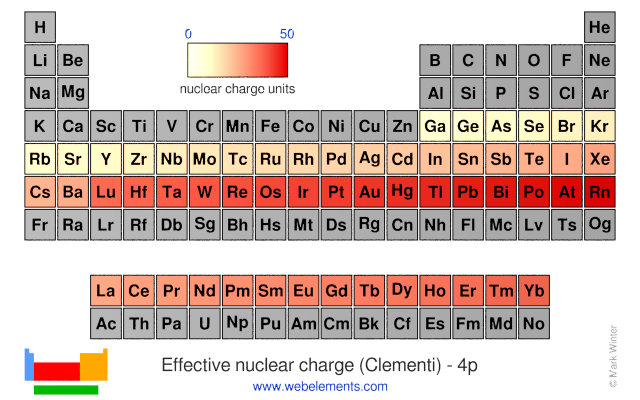
- Highest Zeff: Found in the top-right of the periodic table (excluding noble gases for a moment, as their electron configurations are a bit special, but their Zeff is high!). Think elements like Fluorine (F), Chlorine (Cl), Oxygen (O). These guys are really holding onto their electrons tightly.
- Lowest Zeff: Found in the bottom-left of the periodic table. Think elements like Francium (Fr), Cesium (Cs), Potassium (K). These atoms have their outermost electrons feeling a much weaker pull from the nucleus, making them more likely to lose those electrons. They’re the friendly ones, happy to share!
It's a bit like a heatmap! Hot colors in the top right, cool colors in the bottom left. If you were to draw lines of equal effective nuclear charge, you'd see them generally going diagonally from the bottom left to the top right.
A Little Nuance for the Curious Minds
Now, I know some of you are thinking, "What about the transition metals?" Ah, excellent question! Transition metals are a little trickier because of their partially filled d-orbitals. The shielding provided by d-electrons isn't as effective as that from s and p electrons. This means that across a transition metal series, the effective nuclear charge increases a bit more than you might expect. It's like adding some extra, less effective bodyguards to the mix.

And the noble gases? While they have a full outer shell and are generally considered stable, their effective nuclear charges are actually quite high, especially for the lighter ones like Neon and Argon. This high Zeff is why they are so reluctant to gain or lose electrons. They’re perfectly content with their electron configuration, thank you very much. They’re the ones who always bring snacks to the party but never share.
Why This Matters (Besides Being Super Cool)
So, we’ve ranked them, we’ve chatted about their popularity. But why is this knowledge actually useful? Well, remember what I said about chemical behavior? Effective nuclear charge is the key determinant of things like:
- Atomic Radius: Elements with higher Zeff tend to have smaller atomic radii because the nucleus is pulling those outer electrons in more tightly. Think of it like a stronger magnet pulling on metal filings – they get pulled closer.
- Ionization Energy: This is the energy required to remove an electron. If the effective nuclear charge is high, it means the nucleus has a strong grip on its electrons, so it takes more energy to remove them. The celebrity isn’t letting go of their fans easily!
- Electron Affinity: This is the energy change when an atom gains an electron. Elements with a high Zeff have a strong attraction for electrons, so they tend to have a more negative electron affinity (meaning they readily accept new electrons). They’re actively seeking new fans!
Understanding effective nuclear charge helps us predict how elements will react, what kinds of bonds they will form, and even their physical properties. It’s the underlying engine of the periodic table’s organized chaos!
The Uplifting Finale!
So there you have it! We've journeyed through the world of effective nuclear charge, learned how it changes across periods and down groups, and even had a little peek at the outliers. It’s a concept that might seem a bit abstract at first, but once you grasp it, the periodic table just… clicks. It’s like understanding the secret handshake that explains why everything behaves the way it does.
And the best part? Every atom, from the tiniest Helium to the most massive Uranium, is playing out this dance of nuclear attraction and electron shielding. It’s a constant, dynamic interplay that creates all the amazing diversity of matter we see around us. It’s a reminder that even at the smallest, most fundamental level, there’s order, there are patterns, and there’s a beautiful, predictable rhythm to it all. So next time you look at an element, remember its effective nuclear charge, and smile, because you understand a little piece of its atomic soul. Keep exploring, keep questioning, and never stop finding the wonder in the world around you – it’s truly electrifying!