Ever wondered what happens when you cook an egg? That gooey, clear liquid transforms into that fluffy, solid white, right? Or maybe you’ve seen how heat can change the texture of meat? Well, what you’re witnessing is a fascinating biological process called denaturation. It sounds super scientific, but at its core, it’s all about things losing their natural shape. Think of it like a carefully folded origami crane getting smooshed – it’s still paper, but it’s definitely not a crane anymore!
So, let’s dive into this neat world of denaturation. What exactly is going on under the hood? And more importantly, what are some of the true things about it that might surprise or amuse you? We’re going to break it down, keep it simple, and maybe even have a little fun along the way. Grab a comfy seat, and let’s get curious!
The Shapeshifters of the Cellular World
At the heart of denaturation are proteins. These guys are the workhorses of our bodies, and, indeed, of all living things. They build stuff, they carry things, they send signals – they’re basically the tiny, incredibly busy construction workers and messengers of the cell. And these proteins have really specific, intricate 3D shapes. Think of them like tiny, complex sculptures, each with a job that depends entirely on its precise form.
Imagine a key. Its shape is super specific, right? It only fits into one lock. If you bent that key, it wouldn't work anymore. Proteins are a bit like that. Their job depends on their conformation, which is just a fancy word for their 3D shape. When a protein denatures, it’s like that key getting bent out of shape. It loses its original, functional form.
This losing of shape isn't necessarily a bad thing, at least not always. Sometimes, it's exactly what's needed! Other times, it can be a bit of a disaster for the protein, leading to it becoming… well, useless. So, let’s explore some of the statements that are genuinely true about this whole denaturation game.

Statement 1: Heat is a Common Denaturant.
This one is a biggie, and it’s absolutely true! Remember that egg? Heating it up causes the proteins in the egg white (mostly albumin) to unfold. The heat energy makes the atoms within the protein vibrate more vigorously. This increased jiggling and shaking breaks the relatively weak bonds that hold the protein in its folded, functional shape. It’s like a gentle breeze that, with enough force, can rustle the leaves off a tree.
Think about cooking steak. Raw steak is soft and pliable. Cook it, and it becomes firmer and tougher. That’s because the heat is denaturing the muscle proteins. They’re unfolding and clumping together, changing the texture. So, next time you’re grilling, you're literally performing a chemical reaction! Pretty cool, huh?
Statement 2: Extreme pH Levels Can Also Denature Proteins.
You betcha, this is also true! pH refers to how acidic or alkaline something is. Think of a battery acid versus baking soda. Both are pretty extreme, and so can be the pH environments that proteins find themselves in. Proteins have charged groups on their surface. If the surrounding solution is too acidic (lots of positive hydrogen ions) or too alkaline (lots of negative hydroxide ions), these ions can interact with the protein's charged groups.

These interactions can disrupt the delicate balance of charges within the protein that help maintain its folded structure. Imagine trying to keep a delicate structure of magnets together, but then introducing a whole bunch of other, stronger magnets with opposite charges. They’ll get pulled and pushed out of their original arrangement. This is why strong acids or bases can make proteins change shape.
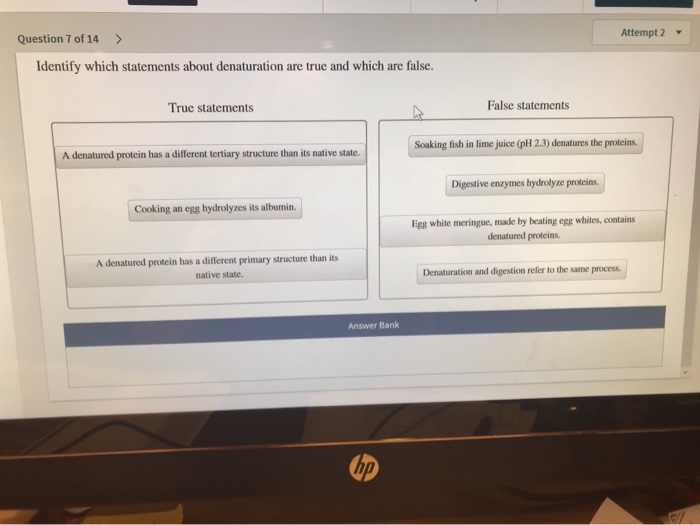
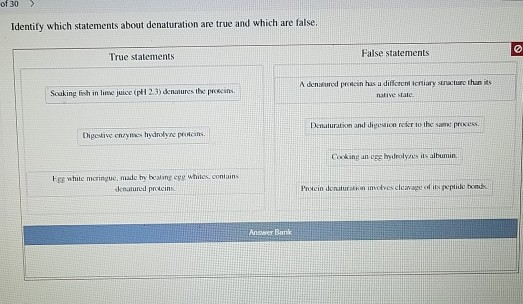
Statement 3: Denaturation is Always Reversible.
Ah, the million-dollar question! And the answer is… false! This is a super important distinction. While some proteins can, under specific conditions, refold themselves after denaturation, it’s absolutely not a universal rule. Think of it like this: some crumpled paper can be smoothed out fairly well. But a paper that’s been soaked in water, torn, and then dried? That’s not going back to its original pristine sheet, is it?
When a protein denatures, especially due to heat or strong chemicals, the unfolding can be so drastic and the resulting clumps so permanent that the original shape is lost forever. The bonds that were broken might not be able to re-form in the correct sequence. So, while some denaturation is reversible, saying all of it is would be a big mistake. The cooked egg white is a perfect example – you can’t uncook it!

Statement 4: Enzymes are Proteins, and Denaturation Affects Their Activity.
Spot on! This statement is unequivocally true. Enzymes are biological catalysts, meaning they speed up chemical reactions in our bodies. And guess what most enzymes are? You guessed it – proteins! Their specific 3D shape is crucial for their function. They have an 'active site,' which is like a perfectly shaped pocket or groove where the molecule they work on (the substrate) fits.
If an enzyme denatures, its active site changes shape. It’s like trying to use a key that’s been bent in a lock. The substrate can no longer fit properly, or at all. This means the enzyme loses its ability to catalyze its reaction. This is why fever can be dangerous; high body temperatures can denature essential enzymes, disrupting vital bodily functions.
Statement 5: Denaturation Can Occur Without Heat or Extreme pH.
Indeed it can! This is another true statement. While heat and extreme pH are common culprits, other factors can also mess with a protein's shape. For instance, mechanical agitation can do it. Think about vigorously whisking egg whites. The physical force of the whisk can cause the proteins to unfold and then re-form into a new, more stable structure, creating those lovely peaks for meringue. It’s like shaking up a snow globe until the snow (protein) settles in a different pattern.

Other chemical agents, like certain salts or organic solvents, can also disrupt protein structure. They can interfere with the water molecules surrounding the protein or directly interact with the protein's amino acid side chains, forcing it to change its shape. So, it’s not just about cooking or using harsh chemicals; sometimes, it’s the sheer force of movement that does the trick.
Why Does This Matter Anyway?
Understanding denaturation isn’t just for scientists in lab coats. It has real-world applications and implications all around us. From the way we prepare food to how our bodies fight off infections, denaturation plays a starring role. For instance, the denaturation of bacterial proteins by disinfectants is how they kill germs. The way our immune system attacks foreign invaders often involves denaturing their proteins.
It’s a reminder that in the microscopic world, shape is everything. The intricate dance of molecules, the precise folding of proteins – it’s all a testament to the complexity and beauty of life. So, the next time you see a cooked egg or even just feel a bit under the weather and wonder why, remember the invisible shapeshifters – the proteins – and their fascinating journey of denaturation. It’s a truly fundamental concept in biology, and a pretty neat one to wrap your head around!